 产品供求
产品供求
- 您当前位置:
- 首页
- 产品数据
- 2,3,4,7,8--五氯二苯并对呋喃
2,3,4,7,8--五氯二苯并对呋喃 基本信息
- 中文名称:
- 2,3,4,7,8--五氯二苯并对呋喃
- 中文别名:
- 2,3,4,7,8-五氯二苯并呋喃;
2,3,4,7,8--五氯二苯并对二恶英
- 英文名称:
- 2,3,4,7,8-PENTACHLORODIBENZOFURAN
- 英文别名:
- 2,3,4,7,8-Pentachlorodibenzofuran;
4-PeCDF;
2,3,4,7,8-P5CDF;
2,3,4,7,8-pentachlorodibenzodioxin;
2,3,4,7,8-heptachlorodibenzofuran;
2,3,4,7,8-pentachlorinated dibenzofuran;
PeCDF;
Dibenzofuran,2,3,4,7,8-pentachloro;
2,3,4,7,8-Pentapolychlorinated dibenzofuran;
2,3,4,7,8-tetrachlorodibenzofuran
- CAS No.:
- 57117-31-4
- 分 子 式:
- C12H3Cl5O
- 分 子 量:
- 340.41662
- 精确分子量:
- 337.86300
- PSA:
- 13.14000
- InChI:
- The Key: OGBQILNBLMPPDP-UHFFFAOYSA-N
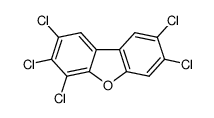
- 分子结构式:
-
- SDS:
- 查看
2,3,4,7,8--五氯二苯并对呋喃 物化性质
- 密度:
- 1.7g/cm3
- 熔点:
- 196-196.5ºC
- 沸点:
- 450.6ºC at 760 mmHg
- 闪点:
- 226.3ºC
- 折射率:
- 1.715
2,3,4,7,8--五氯二苯并对呋喃 毒理性
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- HP5295150
- CHEMICAL NAME :
- Dibenzofuran, 2,3,4,7,8-pentachloro-
- CAS REGISTRY NUMBER :
- 57117-31-4
- LAST UPDATED :
- 199806
- DATA ITEMS CITED :
- 20
- MOLECULAR FORMULA :
- C12-H3-Cl5-O
- MOLECULAR WEIGHT :
- 340.40
- WISWESSER LINE NOTATION :
- T B656 HOJ DG EG FG KG LG
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 916 ug/kg
- TOXIC EFFECTS :
- Behavioral - changes in motor activity (specific assay)
Skin and Appendages - hair
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- FAATDF Fundamental and Applied Toxicology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1981- Volume(issue)/page/year:
11,236,1988
- TYPE OF TEST :
- LDLo - Lowest published lethal dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Primate - monkey
- DOSE/DURATION :
- 34 ug/kg
- TOXIC EFFECTS :
- Endocrine - other changes
Skin and Appendages - hair
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- TXAPA9 Toxicology and Applied Pharmacology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1959- Volume(issue)/page/year:
93,231,1988
- TYPE OF TEST :
- LDLo - Lowest published lethal dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - guinea pig
- DOSE/DURATION :
- 10 ug/kg
- TOXIC EFFECTS :
- Nutritional and Gross Metabolic - body temperature decrease
- REFERENCE :
- ANYAA9 Annals of the New York Academy of Sciences. (New York Academy of
Sciences, 2 E. 63rd St., New York, NY 10021) V.1- 1877-
Volume(issue)/page/year: 320,151,1979
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 91 ug/kg/13W-C
- TOXIC EFFECTS :
- Liver - changes in liver weight
Endocrine - changes in thymus weight
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- CMSHAF Chemosphere. (Pergamon Press Inc., Maxwell House, Fairview Park,
Elmsford, NY 10523) V.1- 1972- Volume(issue)/page/year: 18,265,1989
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1820 ug/kg/13W-C
- TOXIC EFFECTS :
- Liver - changes in liver weight
Endocrine - changes in thymus weight
Blood - changes in leukocyte (WBC) count
- REFERENCE :
- CMSHAF Chemosphere. (Pergamon Press Inc., Maxwell House, Fairview Park,
Elmsford, NY 10523) V.1- 1972- Volume(issue)/page/year: 17,1099,1988
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 360 ug/kg/10W-I
- TOXIC EFFECTS :
- Liver - changes in liver weight
Kidney, Ureter, Bladder - changes in bladder weight
Endocrine - changes in thymus weight
- REFERENCE :
- CMSHAF Chemosphere. (Pergamon Press Inc., Maxwell House, Fairview Park,
Elmsford, NY 10523) V.1- 1972- Volume(issue)/page/year: 20,1165,1990
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 360 ug/kg/12W-I
- TOXIC EFFECTS :
- Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - hepatic microsomal mixed oxidase (dealkylation, hydroxylation,
etc.)
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - transaminases
- REFERENCE :
- CMSHAF Chemosphere. (Pergamon Press Inc., Maxwell House, Fairview Park,
Elmsford, NY 10523) V.1- 1972- Volume(issue)/page/year: 14,953,1985
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 100 ug/kg
- SEX/DURATION :
- female 8 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- FAATDF Fundamental and Applied Toxicology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1981- Volume(issue)/page/year:
12,358,1989
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 30 ug/kg
- SEX/DURATION :
- female 8 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- FAATDF Fundamental and Applied Toxicology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1981- Volume(issue)/page/year:
12,358,1989
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 300 ug/kg
- SEX/DURATION :
- female 8 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
Reproductive - Specific Developmental Abnormalities - respiratory system
Reproductive - Specific Developmental Abnormalities - endocrine system
- REFERENCE :
- FAATDF Fundamental and Applied Toxicology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1981- Volume(issue)/page/year:
12,358,1989
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 4 ug/kg
- SEX/DURATION :
- female 10-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- TXAPA9 Toxicology and Applied Pharmacology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1959- Volume(issue)/page/year:
90,206,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 320 ug/kg
- SEX/DURATION :
- female 10-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - other effects
Endocrine - antidiuresis
Reproductive - Effects on Embryo or Fetus - extra-embryonic structures
(e.g., placenta, umbilical cord)
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 45,611,1992
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 120 ug/kg
- SEX/DURATION :
- female 10-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
- REFERENCE :
- TXAPA9 Toxicology and Applied Pharmacology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1959- Volume(issue)/page/year:
90,206,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 20 ug/kg
- SEX/DURATION :
- female 10-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - urogenital system
- REFERENCE :
- TXAPA9 Toxicology and Applied Pharmacology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1959- Volume(issue)/page/year:
91,246,1987
*** REVIEWS ***
IARC Cancer Review:Animal Limited Evidence
IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals
to Man. (WHO Publications Centre USA, 49 Sheridan Ave., Albany, NY 12210)
V.1- 1972- Volume(issue)/page/year: 69,345,1997
IARC Cancer Review:Human Inadequate Evidence
IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals
to Man. (WHO Publications Centre USA, 49 Sheridan Ave., Albany, NY 12210)
V.1- 1972- Volume(issue)/page/year: 69,345,1997
IARC Cancer Review:Group 3
IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals
to Man. (WHO Publications Centre USA, 49 Sheridan Ave., Albany, NY 12210)
V.1- 1972- Volume(issue)/page/year: 69,345,1997
