 产品供求
产品供求
卓波酰胺 基本信息
- 中文名称:
- 卓波酰胺
- 中文别名:
- 卓波酰胺
- 英文名称:
- Benzamide,3-(aminosulfonyl)-4-chloro-N-[(3aR,4R,7S,7aS)-octahydro-4,7-methano-2H-isoindol-2-yl]-,rel-
- 英文别名:
- 4-Chloro-N-((3aR,4S,7R,7aS)-hexahydro-1H-4,7-methanoisoindol-2(3H)-yl)-3-sulfamoylbenzamide;
Normonal;
Tripamide;
4,7-Methano-2H-isoindole,benzamide deriv.;
E 614;
Toripamide;
ADR 033
- CAS No.:
- 73803-48-2
- 分 子 式:
- C16H20ClN3O3S
- 分 子 量:
- 0
- 精确分子量:
- 369.09100
- PSA:
- 100.88000
- InChI:
- The Key: UHLOVGKIEARANS-UHFFFAOYSA-N
- 安全术语:
- An experimental teratogen. Other experimental reproductive effects. When heated to decomposition it emits toxic fumes of Cl−, SOx and NOx.
- 分子结构式:
-
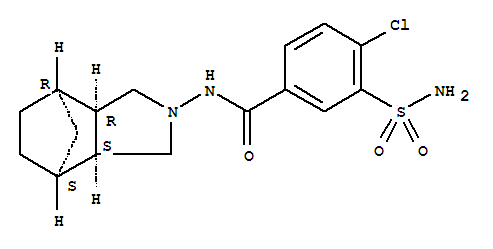
- SDS:
- 查看
卓波酰胺 物化性质
- 密度:
- 1.51g/cm3
- 折射率:
- 1.673
- 存储条件/存储方法:
- 库房通风低温干燥
卓波酰胺 毒理性
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- CV2338880
- CHEMICAL NAME :
- Benzamide,
3-(aminosulfonyl)-4-chloro-N-(octahydro-4,7-methano-2
H-isoindol-2-yl)-
(3a-alpha,4-alpha,7-alpha,7a-alpha)-
- CAS REGISTRY NUMBER :
- 73803-48-2
- BEILSTEIN REFERENCE NO. :
- 1663663
- LAST UPDATED :
- 199612
- DATA ITEMS CITED :
- 13
- MOLECULAR FORMULA :
- C16-H20-Cl-N3-O3-S
- MOLECULAR WEIGHT :
- 369.90
- WISWESSER LINE NOTATION :
- T C555 A ENTJ EMVR DG CSZW
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >8 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >4 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >2 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intramuscular
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >2 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >2500 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intramuscular
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >2500 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- >2 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,APP-15,1982
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 5 gm/kg
- SEX/DURATION :
- female 8-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14,
1968-81. Suspended. Volume(issue)/page/year: 12,651,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 40 gm/kg
- SEX/DURATION :
- female 8-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14,
1968-81. Suspended. Volume(issue)/page/year: 12,651,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 5 gm/kg
- SEX/DURATION :
- female 8-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - physical
- REFERENCE :
- YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14,
1968-81. Suspended. Volume(issue)/page/year: 12,651,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 520 mg/kg
- SEX/DURATION :
- female 7-19 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- YKRYAH Yakubutsu Ryoho. Medicinal Treatment. (Tokyo, Japan) V.1-14,
1968-81. Suspended. Volume(issue)/page/year: 12,669,1979
